Hepatitis Viruses are a group of infectious liver diseases caused by hepatotropic viruses belonging to different families. There are 5 major viruses that cause hepatitis. They make up two groups of hepatitis: enteric (HAV and HEV) and parenteral (HBV, HCV and HDV) (see below table). Enteric hepatitis are characterized by a fecal-oral transmission way and these viruses cause only acute hepatitis. Viruses of enteric hepatitis possess high infectivity and stability. Viruses of hepatitis В, С and D are enveloped in a membrane, are transmitted by parenteral way and are able to promote not only acute but chronic virus hepatitis. Viruses of hepatitis В and С play an important role in the development of chronic virus liver diseases, they are responsible for development of 60-70 percent of hepatic cirrhosis and up to 70-80 percent of primary liver cancers. Due to a great incidence of hepatitis viruses one of the major task is the development of highly sensitive and reproducible methods of diagnostics allowing detection of the causative agent at all stages of the disease as well as monitoring of antiviral therapy effectiveness.
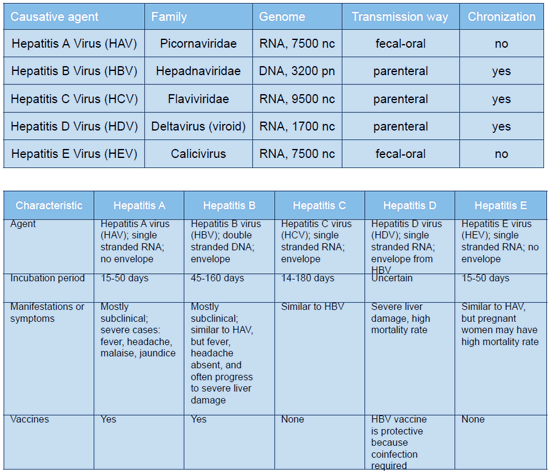
The Hepatitis A (HAV) virus is the enteric infection most widely spread in the world. HAV is one of the five most economically significant infectious diseases and one of the priority problems of the public healthcare. Detection of HAV RNA by PCR method has significant advantages as related to ELISA and biochemical tests at detection of the virus in blood of contact persons as RNA of the hepatitis A virus manifests itself in the blood on the third week from the moment of contamination and is detected at the average within 20 days after appearance of the disease symptoms. Thus, RNA is the first diagnostic marker detected in the patient blood, occurs earlier than a HAV IgM and gives no falsely negative reactions. Detection of HAV RNA with the help of PCR in feces is possible from the third week of the incubation period and up to three months after manifestation of the disease symptoms. Detection of RNA has more advantages (by 1000 times more at the least) as compared to detection of HAV-Ag in environmental entities (drinking or waste waters, waters from impounded surface waters and so on).
| Kit | Description | N. test | Code |
|---|---|---|---|
| HAV Real-TM | |||
| Real Time PCR kit | Real Time Amplification test for detection of Hepatitis A virus (HAV) | 50 | V4-50FRT |
Hepatitis B virus (HBV) is a widely spread human infection caused by DNA-containing virus of HBV belonging to the family Hepadnaviridae. At present in accordance with the WHO data the population infected with HBV virus makes 500 million people. Detection of HBV DNA is used for: – Early diagnostics of acute viral hepatitis B; – Detection of latent forms of viral hepatitis B; – Detection of mutant strains of hepatitis B virus by HBsAg; – Establishment of diagnosis of chronic viral hepatitis B; – Monitoring of effectiveness of the antiviral therapy; ADVANTAGES OF SACACE HBV REAL TIME QUANT KIT – Use of the Quantitative Internal Control (concentration reported in Data Card) which represents recombinant DNA-containing-structure which carried through all steps of analysis from nucleic acid extraction to PCR amplification. The presence of quantitative HBV Rec IC allows not only to monitor the extraction procedure and to check possible PCR inhibition but also to verify possible losses of the DNA during extraction procedure thus enabling to calculate precisely the HBV viral load. – Presence in the reagents supplied with the kit of two positive controls of the extraction: Pos1 HBV low viral load and Pos2 HBV medium viral load that are quantitatively described in Data Card which carried through all steps and allow quality control of the conducted analysis. – High sensitivity: 20 copies/ml (value obtained using the Magno-Virus extraction kit (Sacace REF K-2-16)
| Kit | Description | N. test | Code |
|---|---|---|---|
| HBV Real-TM Quant Dx | |||
| HBV DNA PCR kit (lyophilized, ready to use) | Real Time Amplification CE-IVD test for quantitative detection of HBV DNA with lyophilized and ready to use reagents | 96 | V5-96/3FRT |
| HBV Real-TM Quant new version | |||
| HBV PCR kitLiquid format | Real Time Amplification test for quantitative detection of Hepatitis B virus (HBV) | 100 | V5-100/2FRT |
| HBV Real-TM Qual new version | |||
| HBV PCR kitLiquid format | Real Time Kit for the Qualitative detection of Hepatitis B Virus in human plasma | 100 | V5-100FRT |
Hepatitis C virus (HCV) is RNA-containing, hepatotropic virus belonging to the Flaviviridae family. Contamination with HCV occurs at direct entering of the virus in blood. Most people with acute HCV infection are asymptomatic or have mild symptoms but they are unable to clear the virus and in approximately 80% of cases this leads to chronic infection. In 15 to 20% of patients chronic HCV infection progresses at a variable rate to cirrhosis, with a 1 to 4% annual risk of developing hepatocellular carcinoma. At present there are more than 170 million of infected people, which makes up 3 percent of the population of the world. The leading position in laboratory diagnostics of HCV is taken by molecular-biological methods allowing: 1) early diagnostics of the acute viral infection; 2) establishment of indications to antiviral therapy; 3) choice of the optimum therapeutic regime. ADVANTAGES OF SACACE HCV REAL TIME QUANT KIT – Application of primers and probes in the most conservative area of the UTR region that allow effective detection of the majority of HCV genotypes (tested genotypes: 1a, 1b, 1c, 2a, 2b, 2c, 3a, 3b, 4a, 4c, 4d, 5a, 6a) – Use of the Quantitative Internal Control (concentration reported in Data Card) which represents recombinant RNA-containing-structure which carried through all steps of analysis from nucleic acid extraction to PCR amplification. The presence of quantitative HCV Rec IC allows not only to monitor the extraction procedure and to check possible PCR inhibition but also to verify possible losses of the RNA during extraction procedure thus enabling to calculate precisely the HCV viral load. – Presence in the reagents supplied with the kit of two positive controls of the extraction: Pos1 HCV low viral load and Pos2 HCV medium viral load that are quantitatively described in Data Card which carried through all steps and allow quality control of the conducted analysis – Wide linear range of measurements (from 20 to 50.000.000 IU/ml).
| Kit | Description | N. test | Code |
|---|---|---|---|
| HCV Real-TM Quant Dx | |||
| HCV RNA Quantitative qPCR kit (lyophilized, ready to use) | Real Time Amplification test for quantitative detection of HCV RNA with lyophilized and ready to use reagents | 96 | V1-96/3FRT |
| HCV Real-TM Quant new version | |||
| HCV PCR kitLiquid format | Real Time Amplification test for quantitative detection of Hepatitis C virus (HCV) | 100 | V1-100/2FRT |
| HCV Real-TM Qual new version | |||
| HCV PCR kitLiquid format | Real Time Kit for the Qualitative detection of Hepatitis C Virus in human plasma | 100 | V1-100FRT |
HCV is classified into eleven major genotypes (designated 1-11), many subtypes (designated a, b, c, etc.), and about 100 different strains (numbered 1,2,3, etc.) based on the genomic sequence heterogeneity. Genotypes 1-3 have a worldwide distribution. Types 1a and 1b are the most common, accounting for about 60% of global infections. They predominate in Northern Europe and North America, and in Southern and Eastern Europe and Japan, respectively. Type 2 is less frequently represented than type 1. Type 3 is endemic in south-east Asia and is variably distributed in different countries. Genotype 4 is principally found in the Middle East, Egypt, and central Africa. The determination of the infecting genotype is important for the prediction of response to antiviral treatment: genotype 1 and 4 are generally associated with a poor response to interferon alone, whereas genotypes 2 and 3 are associated with more favourable responses. At patients with subtype 1b the disease progresses to a chronic condition 90 % of cases, in that time as with genotypes 2 and 3b in 33-50 %. In a number of works it is mentioned, that infection with 1b genotype have heavier current of disease with development of a cirrhosis and hepatocarcinoma.
| Kit | Description | N. test | Code |
|---|---|---|---|
| HCV1/2/3 Real-TM Genotype | |||
| Real Time PCR kit | Real Time PCR kit for qualitative detection of and differentiation of hepatitis C virus (HCV) genotypes 1, 2, and 3 RNA in HCV-positive clinical material | 48 | R1-Gen-4x |
| HCV Genotype Plus Real-TM | |||
| Real Time PCR kit | Real-Time test for the determination of HCV-RNA genotypes 1a, 1b, 2, 3, 4, 5a and 6 in the human plasma and simultaneous detection of Internal Control (IC) | 50 | R1-Gen-6 |
| Kit | Description | N. test | Code |
|---|---|---|---|
| HDV Real-TM Quant | |||
| Real Time PCR kit | Real Time PCR Kit for the Quantitative detection of Hepatitis D Virus in human plasma | 120 | V3-100/2FRT |
| HDV Real-TM | |||
| Real Time PCR kit | Real Time Amplification test for detection of Hepatitis D virus (HDV) | 100 | V3-100FRTTV3-100FRTTV3-100FRT C |
| HBV/HDV Real-TM | |||
| Real Time PCR kit | Multiplex Real Time Amplification kit for detection of HBV and HDV | 50 | V56-100FRT |
| Kit | Description | N. test | Code |
|---|---|---|---|
| HGV Real-TM | |||
| Real Time PCR kit | Real Time Amplification test for detection of Hepatitis G virus (HGV) | 50 | V2-50FRT |
The HCV/HBV/HIV Real-TM kit is a qualitative in vitro test for comprehensive single-assay Real Time detec- tion of Human Immunodeficiency Virus (HIV) RNA, hepatitis C Virus RNA and hepatitis B Virus DNA in human plasma with simultaneous detection of Internal Control (IC). This kit is intended for use as a donor screening test to detect HIV RNA, HCV RNA and HBV DNA in plasma from individual donors which may be screened as individual samples or may be tested in pools comprised of equal aliquots.
| Kit | Description | N. test | Code |
|---|---|---|---|
| HCV/HBV/HIV1/HIV2 Real-TM (lyophilized format) | |||
| Real Time PCR kit | Multiplex Real Time amplification kit for detection and differentiation of nucleic acids of HCV RNA, HBV DNA, HIV-1 RNA and HIV-2 RNA (lyophilized format) | 48 | V50-50FRT/L |
| HCV/HBV/HIV1/HIV2 Real-TM (liquid format) | |||
| Real Time PCR kit | Multiplex Real Time amplification kit for detection and differentiation of nucleic acids of HCV, HBV, HIV1 and HIV2. | 100 | V62-100FRT |
Interleukin-28 (IL28) is a cytokine that plays a role in immune defense against viruses. IL28B belongs to the type III interferon family of cytokines. Its classification as interferon is due to its ability to induce an antiviral state. Polymorphisms in the IL28B gene region (rs12979860 and rs8099917) are important in predicting outcome following therapy for chronic hepatitis C virus (HCV) infection. Combined therapy INF pegylated (PEG-IFN) and ribavirin (RBV) is the current standard therapy against HCV infection and to know in detail the polymorphism in IL28B gene region of patients infected with HCV can be an important component of the decision to initiate treatment with PEG-IFN and RBV.
| Kit | Description | N. test | Code |
|---|---|---|---|
| IL28B rs17/rs60 Real-TM | |||
| Real Time PCR kit | Real Time PCR kit for detection of polymorphisms rs8099917 and rs12979860 of Interleukin-28B gene region | 100 | R-O5-100FRT |
Hepatitis B virus (HBV) infects nearly two billion people worldwide. The hepatitis B virus (HBV) is currently categorized into eight genotypes (A to H). Genotypes have been found to be geographically distributed. Numerous studies have investigated the clinical implications of HBV genotypes to disease severity, response to IFN, disease chronicity and hepatocellular carcinoma (HCC).
| Kit | Description | N. test | Code |
|---|---|---|---|
| HBV Genotype A, B, C, D Real-TM | |||
| PCR Real Time kit | Real Time PCR kit for detection and differentiation of hepatitis B virus genotypes A, B, C and D in HBV-positive clinical material | 50 | R5-Gen |
